Search results with tag "Hydroxide"
Student safety sheets 31 Sodium hydroxide
science.cleapss.org.ukPotassium hydroxide is also harmful if swallowed if 3 M or more. Fehling’s solution contains sodium hydroxide of this concentration. It is used in the home as an oven cleaner. Dilute sodium or potassium hydroxide solution (Sodium hydroxide If less than 0.5 M but 0.125 M or more; potassium hydroxide if less than 0.4 M but 0.1 M or more) IRRITANT
SAFETY DATA SHEET SODIUM HYDROXIDE - Bunzl CHS
www.bunzlchs.comSAFETY DATA SHEET SODIUM HYDROXIDE SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product name SODIUM HYDROXIDE Product number 1256 Synonyms; trade names CAUSTIC SODA ANHYDROUS,PEARL,FLAKES,CAUSTIC SODA,SODIUM HYDROXIDE CHEM …
Sodium Hydroxide (NaOH) - Liquiflo
www.liquiflo.comSodium Hydroxide with Liquiflo Gear Pumps Application Note Number: 0101-1 Date: January 18, 2001; Revised Jan. 2016 443 North Avenue, Garwood, NJ 07027, USA Phone: 908-518-0777 • Fax: 908-518-1847 Sodium hydroxide is one of the most commonly used industrial chemicals. It is the third most produced
Right to Know Hazardous Substance Fact Sheet
nj.govCommon Name: SODIUM HYDROXIDE Synonyms: Caustic Soda; Lye; Sodium Hydrate Chemical Name: Sodium Hydroxide Date: April 2010 Revision: November 2015 CAS Number: 1310-73-2 RTK Substance Number: 1706 DOT Number: UN 1823 (solid) UN 1824 (solution) Description and Use Sodium Hydroxide is an odorless, white solid that absorbs
Sodium Hydroxide Solution Safety Data Sheet
nano3dsystems.comSodium Hydroxide Solution Safety Data Sheet Page 1 of 9 Section 1: Identification 1.1 Identification Product Name : Sodium hydroxide solution 1.2 Relevant identified uses of the substance or mixture and uses advised against Use of the substance/mixture : Laboratory chemicals, Plating 1.3 Details of the supplier of the safety data sheet ...
MATERIAL SAFETY DATA SHEET
pgproductsafety.comSODIUM HYDROXIDE 1310-73-2 0.5-1.5 4. First Aid Measures ... US - Illinois Chemical Safety Act: Listed substance SODIUM HYDROXIDE (CAS 1310-73-2) SODIUM HYDROXIDE SODIUM HYPOCHLORITE (CAS 7681-52-9) SODIUM HYPOCHLORITE ... Disclaimer This MSDS is intended to provide a brief summary of our knowledge and guidance regarding the
STANDARDIZATION OF A SODIUM HYDROXIDE SOLUTION …
lshmwaco.orgthe sodium hydroxide solution, there must be a substance used to standardize against. This substance will be potassium hydrogen phthalate (KHP). KHP is an organic acid that is solid and thus, easily weighed. STANDARDIZATION OF A SODIUM HYDROXIDE SOLUTION EXPERIMENT 14. 140
Advanced Water Treatment Concepts
www.wvdhhr.org3. Sodium hydroxide (NaOH): Sodium hydroxide, or caustic soda, may be available in liquid, pellet and flake form. When purchased in liquid form it is easily dispensed to raise pH values in a water system. The liquid will be in a 50% solution of sodium hydroxide and this product is a strong base that should be handled with caution.
KOH Material Safety Data Sheet Chemical Name: Potassium ...
www.uni-muenster.deMaterial Safety Data Sheet Potassium hydroxide MSDS Section 1: Chemical Product and Company Identification Product Name: Potassium hydroxide ... trichloride, peroxidized tetrahydrofuran, 2,4,6-trinitrotoluene. Reaction with ammonium hexachloroplatiate(2-) + heat forms heat sensitive explosive product. Potassium hydroxide will cause explosive ...
EXPERIMENT 12 A: STANDARDIZATION OF A SODIUM …
www.cerritos.eduSodium hydroxide solution of about 0.2 M is prepared in order to be used in Exp 12B. The solution is then standardized, that is, its concentration is accurately determined, by titrating it against a weighed sample of known acidic salt, potassium acid phthalate. INTRODUCTION: Sodium hydroxide is deliquescent (absorbs moisture from the atmosphere ...
Standardization of a NaOH Solution with Potassium …
mymission.lamission.edusodium hydroxide solution. Although sodium hydroxide is a solid, preparation of solutions of accurately known concentration is difficult. The base is very hygroscopic, water absorbing. Additionally, the resulting solution tends to absorb carbon dioxide from the air, which neutralizes some of the base. Hence, we first standardize the base
Right to Know Hazardous Substance Fact Sheet
nj.govThe following chronic (long-term) health effects can occur at some time after exposure to Potassium Hydroxide and can last for months or years: Cancer Hazard ... moisture or water may generate enough heat to ignite combustibles (wood, paper and oil). POTASSIUM HYDROXIDE Page 4 of 6 For more information, please contact: New Jersey Department of ...
Core practical 2: Find the concentration of a solution of ...
qualifications.pearson.comCore practical 2Technician sheet Find the concentration of a solution of sodium hydroxide Practical activities have been safety checked but not trialled by CLEAPSS. Users may need to adapt the risk assessment information to local circumstances. Core practical 2: Find the concentration of a solution of sodium hydroxide Objective
Sodium Hydroxide - labchem.com
www.labchem.comSodium Hydroxide Safety Data Sheet according to Federal Register / Vol. 77, No. 58 / Monday, March 26, 2012 / Rules and Regulations Date of issue: 07/06/1998 Revision date: 02/21/2018 Supersedes: 10/14/2013 Version: 1.1
物質安全資料表 - THU
gclab.thu.edu.tw中英文名稱:氫氧化鈉(Sodium hydroxide) 同義名稱:苛性鈉、燒鹼、 Caustic soda 、 Caustic flake 、 LYE 、 Soda lye 、 Sodium hydrate 、 White caustic 、 Hydroxyde de sodium (solide) 、 Sodium hydroxide . 化學文摘社登記號碼( CAS No. ): 1310-73-2 . 危害物質成分(成分百分比): 四、 急救 ...
Material Safety Data Sheet (MSDS) – Caustic Soda (NaOH)
www.itokindo.orgSodium Hydroxide Technical Grade, Flake Nama Dagang Formula: NaOH RTECS: WB4900000 C.A.S CAS# 1310-73-2 Bagian 2. Komposisi Sara 313 Komponen Nomor CAS % Dim Batas penggunaan: Sodium Hydroxide CAS# 1310-73-2 100.00% W/W OSHA PEL 2 mg/mƒ ACGIH 2mg/mƒ Bagian 3. Pengenalan Bahaya Parah menyebabkan iritasi dan luka bakar. Berbahaya …
TITRATION OF SULPHURIC ACID WITH SODIUM HYDROXIDE
wikieducator.orgNov 03, 2002 · Finally, the sodium hydroxide in the burette is added to the acid/indicator solution until the indicator changes colour. (An indicator dye is a chemical that has a different colour in an acid and a base.) PROCEDURE 1. Get the following equipment for your group: burette pipette syringe with piece of rubber tubing attached 1– 250 mL beaker
Sodium Hydroxide, 1.0N (1.0M)
www.labchem.comSodium Hydroxide, 1.0N (1.0M) Safety Data Sheet according to Federal Register / Vol. 77, No. 58 / Monday, March 26, 2012 / Rules and Regulations 07/22/2020 EN (English US) 5/9 Solubility : Soluble in water. Log Pow : No data available Auto-ignition temperature : No data available Decomposition temperature : No data available
SAFETY DATA SHEET (SDS)
www.tatachemicals.comJun 21, 2021 · Hydroxide Common Name Sodium Hydroxide Synonyms Caustic Soda, Soda Lye, Lye Company Name / Address / Phone / Fax Tata Chemicals Limited, Mithapur District: Devbhoomi Dwarka (Gujarat) 361 345 INDIA Contact in case of Emergency Only: +91(02892 67 5802 /03 ) Contact Person :+91 9227676113 Mr. Devendra Thakur Chemical Name Sodium …
Reaction of Aluminum with Water to Produce Hydrogen
www1.eere.energy.govThe first reaction forms the aluminum hydroxide bayerite (Al(OH) 3) and hydrogen, the second reaction forms the aluminum hydroxide boehmite (AlO(OH)) and hydrogen, and the third reaction forms aluminum oxide and hydrogen. All these reactions are thermodynamically favorable from room temperature past the melting point of aluminum (660 . o. C).
Ionic Compound Formula Writing Worksheet
www.spart5.netsodium carbonate 2) NaOH sodium hydroxide 3) MgBr 2 magnesium bromide 4) KCl potassium chloride 5) FeCl 2 iron (II) chloride 6) FeCl 3 iron (III) chloride 7) Zn(OH) 2 zinc hydroxide 8) Be 2 SO 4 beryllium sulfate 9) CrF 2 chromium (II) fluoride 10) Al 2 S 3 aluminum sulfide 11) PbO lead (II) oxide 12) Li 3 PO
Sodium Hydroxide, 5% w/v
www.labchem.comSodium Hydroxide, 5% w/v Safety Data Sheet according to Federal Register / Vol. 77, No. 58 / Monday, March 26, 2012 / Rules and Regulations 11/06/2020 EN (English US) 2/9
Expt 3: Reduction of Vanillin with Sodium Borohydride to ...
tigerweb.towson.edufirst be deprotonated with the strong base sodium hydroxide in order for it to dissolve. Phenols (pKa = 10) are considerably more acidic than ordinary alcohols (pKa = 16-18) because the conjugate base (called a phenolate) is resonance stabilized. Since sodium hydroxide is the conjugate base of water (pK a = 15.7), the deprotonation of vanillin will
Form and Stability of Aluminum Hydroxide Complexes in ...
pubs.usgs.govAluminum has a particularly strong tendency to hydrolize in solution, and at any pH above 3.5 various combinations of aluminum with hydroxide ions occur. The purpose of this investigation was to determine the effect of pH on aluminum solubility and …
LABORATORY 5 Extraction - Stockton University
blogs.stockton.eduthe flask. Add 10 mL of a 1 M sodium hydroxide solution to the funnel. Stopper the funnel and carefully mix the contents. Vent the funnel and then shake the mixture thoroughly, venting often. Allow the layers to separate completely, remove the stopper from the funnel, and then draw off the lower layer into a labeled 50 mL Erlenmeyer flask.
SAFETY DATA SHEET: TETRAHYDROFURAN
www.ciscochem.comReacts explosively with lithium-aluminum alloys, and Sodium Aluminum Hydride, Potassium hydroxide, Calcium Hydride. It is normally stable, however, prolonged storage, and exposure to air and light may cause formation of unstable explosive peroxides especially when anhydrous and unless it is inhibited against peroxide formation.
DAWN PROFESSIONAL DISH DETERGENT MTR PGNG EN
lockridgepkg.comSodium hydroxide 1310-73-2 1000 lb - - X California Proposition 65 This product is not subject to warning labeling under California Proposition 65. U.S. State Regulations (RTK) Chemical Name CAS-No New Jersey Ethanol 64-17-5 X Page 6 / 7 96663951_PROF_NG-Dawn Professional Dish Detergent Revision Date: 02-Jul-2015
Electrolysis of dilute sulfuric acid - Physics
physics.williams.eduhydrogen and oxygen atoms i.e. by splitting liquid water into two gaseous element molecules of hydrogen (H2) and oxygen (O2). Water only ionises to a tiny extent giving minute concentrations of hydrogen ions and hydroxide ions, so the presence of high concentrations of hydrogen ions (H+ or H 3O +) and sulfate ions (SO4
Buffer Solution pH 10 Safety Data Sheet - Lewis University
www.lewisu.eduSodium Hydroxide 1310-73-2 No 1000 lb RQ 1000lb (454kg) final RQ No No Section 16 Additional Information Revised: 09/09/2015 Replaces: 05/26/2015 Printed: 10-29-2015 The information provided in this (Material) Safety Data Sheet represents a compilation of data drawn directly from various sources available to us.
MATERIAL SAFETY DATA SHEET AMMONIA SOLUTION
www.mysoreammonia.comCHEMICAL NAME: AMMONIUM HYDROXIDE COMPOSITION: 25% by weight of Ammonia gas in Water CHEMICAL FAMILY: Ammonia FORMULA: NH 4 0H or NH 3 (Aq) MOLAR MASS: 35.04 g/mol SYNONYMS: Aqua Ammonia, Aqueous Ammonia, Liquor Ammonia, Liquour Ammonia, Ammoniacal Liquor, Ammonia Water and Ammonia Solution 2. Hazards Identification
SAFETY DATA SHEET - AC Cleaning
www.accleaning.com.ausodium hydroxide NOHSC (2005-01-18) TWA 2 mg/m3 Notes: Peak limitation sodium hypochlorite, solution 95% Cl active EH40-WEL (2007-10-01) STEL 1,5 mg/m3, 0,5 ppm To assure Safety and Healthy workplace OELs are listed in …
Over-the-Counter Drug List - Kentucky
chfs.ky.govDocusate sodium 50 mg/15ml liquid Docusate sodium 60 mg/15ml syrup Docusate sodium 150 mg/15ml liquid Magnesium citrate solution Magnesium hydroxide 400 mg/5ml suspension Sennosides 8.8 mg/5ml syrup Simethicone 40 mg/0.6ml suspension Powders Drug Name Strength Dosage Form Polyethylene glycol 3350 oral 17 gm packet
MATERIAL SAFETY DATA SHEET BLEACH - Lavo
lavo.caToxicological data: There is no available data for the product itself, only for the ingredients. See below for individual ingredient acute toxicity data. Ingredients CAS CL50(4hr) inh, rat DL50 (Oral, rat) DL50 (Rabbit, dermal) Sodium Hydroxide 1310-73-2
Determination of Calcium Ion Concentration
www.canterbury.ac.nzhydroxide solution used is highly corrosive and should be handled with extra care: ideally wear rubber gloves when preparing and handling it. Introduction This method, called a complexometric titration, is used to find the calcium content of milk, the ‘hardness’ of water and the amount of calcium carbonate in various solid materials.
Concentration Worksheet W 328 - Everett Community College
www.everettcc.edu7) Find the volume of a 0.75 M solution if it contains 39 grams of potassium hydroxide. 8) How many grams of hydrochloric acid are present in 3.0 L of a 0.750 M solution? 9) The concentration of oxygen in water at the bottom of a lake is 0.48 g/L and the pressure is 2.5 atm. If water from the bottom is moved by a current upwards to
ALUMINUM FACT SHEET - Water Quality Association
www.wqa.orgAluminum can be selectively leached from rock and soil to enter any water source. Al3+ is known to exist in groundwater in concentrations ranging from 0.1 ppm to 8.0 ppm. Aluminum can be present as aluminum hydroxide, a residual from the municipal feeding of alum (aluminum sulfate) or as sodium aluminate from clarification or precipitation ...
Lysol Brand Concentrate Disinfectant Safety Data Sheet
app.aceworldwide.comSafety Data Sheet Lysol Brand Concentrate Disinfectant Page 1 of 5 ... Potassium Hydroxide 1310-58-3 0.38 Section 4 First Aid Measures Emergency and First Aid Procedures ... Red Liquid Vapor Density (Air=1): No data available Odor: No data …
Student safety sheets 33 Sodium, Potassium, Magnesium ...
science.cleapss.org.ukCalcium & Magnesium carbonate limestone. It Magnesium carbonate hydroxide (‘light’ and ‘heavy’ magnesium carbonate) Solid . LOW HAZARD. Calcium carbonate occurs naturally as calcite, marble, chalk and decomposes if heated above 800°C and gives carbon dioxide with acids (unless the calcium salt is insoluble in water). It is an
Material Safety Data Sheet Product Name: Caustic Soda Flakes
chemfabalkalis.comProduct Identifier: CAUSTIC SODA FLAKES (98%) Caustic soda Flake Sodium hydroxide Flakes Caustic Soda Flakes CAS #: 1310 -73 -2 UN # : 1823 Sodium Carbonate CAS #497-19-8, < 1.0% Formula: NaOH Molecular mass: 40.0 g/mol SECTION 1. Product company identification Emergency Contact number: 0413- 2655111-113 Email: chemfabalkalis@drraoholings.com
What is MMS and How to Make it?
www.mmsinfo.orgreduced with Hydrogen Peroxide. The result is Sodium Chlorite. What are the inert ingredients in Sodium Chlorite The inert ingredients in any given batch of Sodium Chlorite can vary, but will usually be comprised of salts such as Sodium Chloride, Sodium Carbonate, Sodium Sulfate, Sodium Chlorate, and Sodium Hydroxide.
Dawn Ultra Dishwashing Liquid, Original Scent Revision Date
sds.chemtel.netSodium hydroxide 1310-73-2 1000 lb - Clean Air Act, Section 112 Hazardous Air Pollutants (HAPs) (see 40 CFR 61) This product contains the following substance(s) which are either listed as hazardous air pollutants (HAPS) or VOC's per the Clean
OTC Products Catalog - Marketing Catalog Site
catalogcontent.medline.comOTC009185 Aluminum Hydroxide Antacid SF Gel 16oz 16 oz Amphojel ... OTC093616 Docusate Sodium 50 mg/5 ml Liquid 16-oz. 16 oz Colace Liquid OTC40116 Docusate Sodium 60 mg/15 ml Syrup 16-oz. 16 oz Colace Syrup Fiber Item No. Description Pkg. Compare To
Acrylic Modified Alkyd Enamel ALK-200
custompaintusa.com10% NaOH (Sodium Hydroxide) Pass Stain, Wrinkle, Color Change 10% HCl (Hydrochloric acid) Pass Pass 10% H 2 SO 4 ... Material Safety Data Sheets for the PPG products mentioned in this publication are available through your PPG Distributor. For additional information regarding this product, see the MSDS AND LABEL information. ...
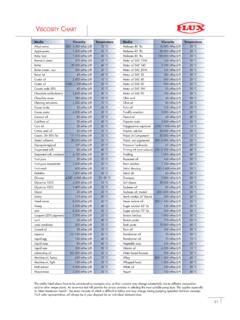
VISCOSITY CHART - Weebly
thesuccesstechnic.weebly.comPolymer solution 20.000 mPas (cP) 20 °C Polyol (A-Component) 85.000 mPas (cP) 10 °C Polyol, non-pigmented 500–5.000 mPas (cP) 20 °C Potassium hydroxide 67 mPas (cP) 20 °C Printing ink (and colours) 550–2.200 mPas (cP) 40 °C Pudding 1.000 mPas (cP) 40 °C Rapeseed oil 160 mPas (cP) 20 °C Resin solution 7.100 mPas (cP) 20 °C
Sodium Hydroxide ENVIRONMENT, SAFETY, HEALTH, AND …
www-group.slac.stanford.eduIncompatible with water, acids, flammable liquids, organic halogens, metals such as aluminum, tin, and zinc, and nitromethane. The substance is a strong base; it reacts violently with acid and is corrosive in moist air to metals like zinc, aluminum, tin, …
FABULOSO ALL PURPOSE CLEANER LIQUID LAVENDER
www.htproducts.netFABULOSO ALL PURPOSE CLEANER LIQUID LAVENDER This industrial Safety Data Sheet is not intended for consumers and does not address consumer use of the product. For information regarding consumer applications of this product, refer to the product label. ... SODIUM HYDROXIDE 1310-73-2 . NA .
Similar queries
SODIUM HYDROXIDE, Hydroxide, Sodium, SAFETY DATA SHEET SODIUM HYDROXIDE, Sodium Hydroxide Solution Safety Data Sheet, SODIUM HYDROXIDE SOLUTION, Safety Data Sheet, Material Safety Data Sheet, Safety, SODIUM HYDROXIDE SODIUM, Summary, LIQUID, Material Safety Data Sheet Potassium hydroxide MSDS, Tetrahydrofuran, Solution, Hydroxide solution, Health effects, Potassium Hydroxide, Heat, Sheet, Flake, Aluminum, Aluminum hydroxide, Ionic Compound Formula Writing Worksheet, Vanillin, Aluminum solubility, SAFETY DATA SHEET: TETRAHYDROFURAN, Electrolysis of dilute sulfuric acid, Data, Kentucky, SAFETY DATA SHEET BLEACH, Calcium, Everett Community College, Flake Sodium hydroxide, ALK-200, Safety Data, VISCOSITY