Ceftriaxone – safety in neonates
therefore currently recommended as first line therapy for gonococcal ophthalmia neonatorum and other gonococcal infections in the new born[11].
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
INVESTING IN HEALTH FOR ECONOMIC …
www.who.int2 INVESTING IN HEALTH FOR ECONOMIC DEVELOPMENT Report by the Mexican Commission on Macroeconomics and health 3 The …
Macroeconomics, Investing in health for economic, Investing, Health, Economic, Investing in health for economic development, Development
WHO GOOD PRACTICES FOR PHARMACEUTICAL …
www.who.intworking document qas/09.297/rev.2 page 2 schedule for the proposed adoption process of document qas/09.297/rev.2: who good practices for pharmaceutical microbiology laboratories
Microbiology, Good practices for pharmaceutical, Good, Practices, Pharmaceutical, Good practices for pharmaceutical microbiology laboratories, Laboratories
NORMES ET STANDARDS EN INFRASTRUCTURES …
www.who.intnormes et standards en infrastructures et equipements du centre de sante et de promotion sociale, du centre medical avec antenne chirurgicale et du …
Heterotrophic Plate Count Measurement in …
www.who.intWorld Health Organization Sustainable Development and Healthy Environments Heterotrophic Plate Count Measurement in Drinking Water Safety Management
Health, Heterotrophic plate count measurement in, Heterotrophic, Plate, Count, Measurement, World health organization, World, Organization
Annex 3 WHO good manufacturing practices for ...
www.who.int96 Introduction The fi rst WHO draft text on good manufacturing practices (GMP) was prepared in 1967 by a group of consultants at the request of the Twentieth
Annex 5 WHO good distribution practices for …
www.who.int236 1. Introduction Distribution is an important activity in the integrated supply-chain management of pharmaceutical products. Various people and entities are
Good, Practices, Pharmaceutical, Good distribution practices for, Distribution
Annex 2 WHO good practices for pharmaceutical …
www.who.int71 Introduction and scope of document Pharmaceutical microbiology laboratories may be involved in: — sterility testing; — detection, isolation, enumeration and identifi cation of microorganisms
Microbiology, Good practices for pharmaceutical, Good, Practices, Pharmaceutical, Laboratories, Pharmaceutical microbiology laboratories
LABORATORY BIOSAFETY MANUAL - WHO
www.who.intinterim guidelines WHO/CDS/CSR/LYO/2003.4 LABORATORY BIOSAFETY MANUAL SECOND EDITION (REVISED) WORLD HEALTH ORGANIZATION Geneva 2003
Health, Manual, Laboratory, World health organization, World, Organization, Laboratory biosafety manual, Biosafety
Mycobacteriology Laboratory Manual - WHO
www.who.intMycobacteriology Laboratory Manual ii Edita Pimkina, Infectious Diseases and Tuberculosis Hospital Affi liate of Public institution …
Manual, Laboratory, Mycobacteriology laboratory manual, Mycobacteriology
Laboratory Guidelines for enumerating CD4 T …
www.who.intSEA-HLM-392 Distribution: Limited Laboratory Guidelines for enumerating CD4 T Lymphocytes in the context of HIV/AIDS Regional Office for South-East Asia
Guidelines, Laboratory, Laboratory guidelines for enumerating cd4, Enumerating
Related documents
The Ontario Curriculum, Grades 11 and 12: Science, 2008 ...
www.edu.gov.on.caIntRoDUCtIon This document replaces The Ontario Curriculum, Grades 11 and 12: Science, 2000.Beginning in September 2009, all science programs for Grades 11 and 12 will be based on the expecta-tions outlined in this document.
2015 REVISED The Ontario Curriculum Grades 11 and 12
www.edu.gov.on.ca5. PREFACE. This document replaces . The Ontario Curriculum, Grades 11 and 12: Canadian and World Studies, 2005. Beginning in September 2015, all Canadian and world studies courses for
Grade, Curriculum, Ontario, Ontario curriculum, Grades 11 and 12, Ontario curriculum grades 11 and 12
Language Arts Florida Standards (LAFS) Grades 11 & 12
www.fldoe.orgCluster 4: Range of Reading and Level of Text Complexity STANDARD CODE STANDARD LAFS.1112.RI.4.10 By the end of grade 11, read and comprehend literary nonfiction in the grades
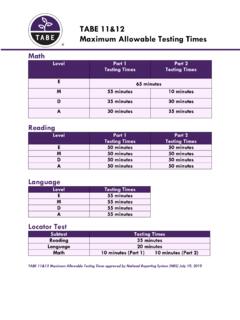
TABE 11&12 Maximum Allowable Testing Times - tabetest.com
www.tabetest.comMath Level Part 1 Testing Times Part 2 Testing Times E M 55 minutes 10 minutes D 35 minutes 30 minutes A 30 minutes 35 minutes Reading
TABE 11 & 12 Scoring Levels: Best Practice Guidance
tabetest.comEach of the TABE test levels (L, E, M, D, and A) has content aligned specifically to a range of NRS levels and a student can only be classified into those NRS levels.
BULLETIN - nema.org
www.nema.org1 engineering department bulletin no. 71 june 1965 revised september 9, 2004 (reaffirmed 12/2011) knockout diameters and fitting dimensions to assure mechanical and electrical continuity
AU-11:Form AU-11:12/10: Application for Credit or Refund ...
www.tax.ny.govAU-11 (12/10) New York State Department of Taxation and Finance Application for Credit or Refund of Sales or Use Tax • Do not use this form to apply for a Qualified Empire Zone Enterprise (QEZE) refund or credit for purchases made on or after September 1, 2009. Use Form AU-12, Application for Credit or Refund of Sales or Use Tax - Qualified Empire Zone Enterprise
Finance, York, Department, States, Applications, Direct, Taxation, Refund, 10 12, Application for credit or refund, New york state department of taxation and finance application for credit or refund
11.12 Concrete Batching - US EPA
www3.epa.gov11.12-26/06 Types of controls used may include water sprays, enclosures, hoods, curtains, shrouds, movable and telescoping chutes, central duct collection systems, and the like.
Related search queries
Ontario Curriculum Grades 11 and 12, Ontario Curriculum, Grades 11 and 12, 11 & 12, LAFS.1112, 11&12, 11:12/10: Application for Credit or Refund, 11 12, New York State Department of Taxation and Finance Application for Credit or Refund, Application for Credit or Refund, 11.12 Concrete Batching, 11.12