Experiment 6 Qualitative Tests for Alcohols, Alcohol ...
alcohol but is specific for only one class of alcohol. This is the secondary methyl alcohol. If the alcohol contains a methyl group attached to a carbon that also has a hydrogen and an OH group then it will give a positive iodoform test. The formation of a yellow precipitate indicates a positive test. This is shown in figure 6.4. R C OH H CH 3 ...
Tags:
Alcohols, Methyl, Methyl alcohol
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
Experiment 17 Preparation of Methyl Orange
myweb.liu.eduFilter the crystals by suction filtration, rinse them with 10 – 15 ml cold water and allow them to dry. Calculate the percent yield and turn in your crystals in a vial along with your Organic Yield Report Sheet. Do not attempt to take the melting point of …
EXPERIMENT 6 Bases, Acids and Antacids - Home | …
myweb.liu.eduEXPERIMENT 6 Bases, Acids and Antacids ... The terms acid and base are used quite loosely in chemistry and ... Vinegar is a product of the aerobic fermentation of ...
Base, Chemistry, Acid, Experiment, Fermentation, Experiment 6 bases, Acids and antacids, Antacids
Experiment 14 Preparation of p-Nitroaniline
myweb.liu.eduDisconnect from the vacuum, wash the solid crystals with 10 mL of cold water and reconnect the vacuum tube for a couple of minutes more so as to dry the product as much as possible. Transfer the crystals to a watch glass and leave them to dry in your locker until the next laboratory session. Weigh the product and record the number in your notebook.
Some Reactions of Hydrocarbons
myweb.liu.eduReactions of Hydrocarbons Experiment #2 Pre-Lab Exercise 1. How would you describe the difference between saturated and unsaturated hydrocarbons and what is a distinguishing feature of aromatic hydrocarbons. 2. Describe the relative reactivity of alkanes, alkenes and aromatic hydrocarbons with respect to their chemical reactions.
EXPERIMENT 11 The Chemistry of Lipids
myweb.liu.eduLipids, by definition, are natural substances that do not mix with water but dissolve in organic solvents. There are several classes of lipids, including: fatty acids, waxes, triacylglycerols (fats and oils), phospholipids and steroids. The fatty acids are usually not free in
The Chemistry of Lipids Experiment #8
myweb.liu.eduThe Chemistry of Lipids Experiment #8 Objective To observe the solubility of lipids in polar and nonpolar solvents and to compare saturated and unsaturated fats in their chemical reaction with bromine. The percent fat in a food will be determined by extraction of the fat and weighing it. …
Experiment 16 Qualitative Analysis of Amines and Amine …
myweb.liu.edu4 (blue to Congo Red). At this point you should see a precipitate. Repeat the benzene sulfonyl chloride treatment using a secondary amine and a tertiary amine. With the secondary amine you should see two layers. Draw off the lower aqueous layer with a separatory funnel and see if the organic layer is soluble in 5% HCl.
Experiment 13 Qualitative Tests for Carbonyls; Unknown ...
myweb.liu.educomplexed with ammonia, to Ag0 (metallic silver) while it is oxidized to a carboxylic acid. Ketones do not undergo this reaction. When the metallic Ag0 is produced it forms a silver mirror on the inner side of the test tube. In order for the mirror to form, the test tube must be very clean and without many scratches inside. The test tubes must be
Experiment 7 Preparation of 1-Bromobutane
myweb.liu.eduThis is an S N 2 reaction. The mechanism is shown in Figure 7.1. The purpose of the sulfuric acid is to first protonate the weakly basic hydroxyl group and thereby convert it into a good leaving group. It can then leave as neutral water. CH 3 CH 2 CH 2 CH 2 OH CH 3CH 2CH 2CH 2 H H O H O S O OH O CH 3 CH 2 CH 2 CH 2 Br CH 3CH 2CH 2CH 2 O H H H ...
Related documents
Methanol
www.labchem.comSynonyms : acetone alcohol / alcohol C1 / alcohol, methyl / carbinol / colonial spirits / columbian spirits / green wood spirits / manhattan spirits / methyl alcohol / methyl hydrate / methyl hydroxide / methylen / methylol / monohydroxymethane / pyroligneous spirit / pyroxylic spirit / wood alcohol / wood naphtha . 1.2.
SAFETY DATA SHEET 1 Denatured Alcohol
www1.mscdirect.comOct 04, 2012 · Ethyl alcohol {Ethanol} 64-17-5 CAS # 40.0 -50.0 % Concentration 1. Methanol {Methyl alcohol; Carbinol; Wood alcohol} 2. 67-56-1 50.0 -55.0 % Methyl isobutyl ketone {Hexone; Isopropylacetone; MIBK; 4-Methyl-2-pentanone} 3. 108-10-1 1.0 -4.0 % 4. Acetic acid, ethyl ester {Ethyl acetate} 141-78-6 0.5 -1.5 % 5. Heptane 142-82-5 0.5 -1.5 % 4. First ...
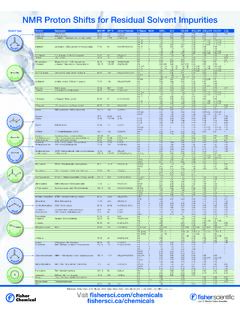
NMR Proton Shifts for Residual Solvent Impurities
beta-static.fishersci.comMethanol Methyl alcohol 32.04 64.7 CH 3OH CH 3 s 3.49 3.34 3.34 3.16 3.31 3.28 3.07 Pentanol n-Amyl alcohol / Pentyl alcohol 88.15 137-139 CH …
Impurities, Alcohols, Residual, Shifts, Methyl, Solvents, Pronto, Methyl alcohol, Proton shifts for residual solvent impurities
Incompatibilities of concentrated nitric acid: Never mix ...
ccc.chem.pitt.eduMethyl bromide Methyl chloride Methylene chloride Monochlorodifluoromethane Perchloroethylene Propylene dichloride 1,2,4-Trichlorobenzene 1,1,1-Trichloroethane Trichloroethylene Trichlorofluoromethane Group 6: Alcohols, Glycols and Glycol Ethers Allyl alcohol Amyl alcohol 1,4-Butanediol Butyl alcohol (iso, n, sec, tert) Butylene glycol Corn syrup
Acid, Alcohols, Methyl, Concentrated, Nitric, Incompatibilities of concentrated nitric acid, Incompatibilities
Q3C (R8): Impurities: guideline for residual solvents
www.ema.europa.euPDE for 2-Methyltetrahydrofuran, Cyclopentyl Methyl Ether, and Tertiary-Butyl Alcohol 4 76 CYCLOPENTYL METHYL ETHER Introduction77 78 Cyclopentyl methyl ether (CPME: CAS Number 5614-37-9) is used in pharmaceutical chemical 79 development as an alternative to its more common analogues such as tetrahydrofuran and tert-butyl 80 methyl ether (1,2).
MATERIAL SAFETY DATA SHEET- ( MSDS ) METHYL ETHYL …
www.pure-chemical.comMETHYL ETHYL KETONE 78 -93 -3 100% * All concentrations are percent by weight unless material is a g as. Gas concentrations are in percent by volume. 3. HAZARDS IDENTIFICATION This material is considered to be hazardous according to regulat ory guidelines (see MSDS Section 15). POTENTIAL PHYSICAL / CHEMICAL EFFECTS Flammable.