Experiment 3: Acid/base Extraction and Separation of ...
of the stronger acid versus the weaker acid. The weaker base, sodium bicarbonate, is strong enough to react with the stronger acid, benzoic acid, but not strong enough to react with the weaker acid, 2-naphthol. The sodium salt that forms is …
Tags:
Base, Salt, Acid, Extraction, Base extraction
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
Expt 3: Reduction of Vanillin with Sodium Borohydride to ...
tigerweb.towson.edufirst be deprotonated with the strong base sodium hydroxide in order for it to dissolve. Phenols (pKa = 10) are considerably more acidic than ordinary alcohols (pKa = 16-18) because the conjugate base (called a phenolate) is resonance stabilized. Since sodium hydroxide is the conjugate base of water (pK a = 15.7), the deprotonation of vanillin will
Experiment 3: Acid/base Extraction and Separation of ...
tigerweb.towson.edulayer separation. Be sure to pick an iron ring that is properly sized - too big and the iron ring will let the separatory funnel fall straight through and break on the counter! First, place your sep funnel in the iron ring, check that the stopcock is SHUT, then add the organic and aqueous solutions. Now, place the stopper on top.
Experiment 11: Dehydration of Cyclohexanol
tigerweb.towson.eduto the funnel. (This process is called “washing”.) The NaOH removes any trace of acid that may have co-distilled with the product. The aqueous layer is carefully removed, leaving the product in the funnel. The “wet” product is then transferred to a …
STRUCTURE DETERMINATION PROBLEMS USING …
tigerweb.towson.eduSTRUCTURE DETERMINATION PROBLEMS USING IR SPECTROSCOPY The IR spectra (A – F) of the six compounds are provided on the following pages. Each of the spectra is produced by one of 17 compounds that are shown below.
Using, Structure, Problem, Precast, Determination, Spectroscopy, Structure determination problems using, Structure determination problems using ir spectroscopy
Herbert J. Gans. The Uses of Poverty: The Poor Pay All.
tigerweb.towson.eduSecond, because the poor are required to work at low wages, they subsidize a var iety of economic activities that benefit the affluent. For example, domestics subsidize the upper middle and upper
Uses, Poverty, Poor, Herbert, Uses of poverty, The poor pay all
Expt 9: The Aldol Condensation - Towson University
tigerweb.towson.edusolids in the vial with a microspatula to make them easier to transfer. Pour the contents of the vial onto the Hirsch funnel and filter the solid. 9. To ensure complete transfer of all the solids into the Hirsch funnel, wash out the conical vial twice, using two 3 mL portions of DI water, transferring all washings into the Hirsch funnel each time.
Reactions of Alkyl Halides - Towson University
tigerweb.towson.edugroup, alleviates both the electronic and steric issues seen in the frontside attack. This is a one-step concerted (i.e. happens all at once) process where all the bond formations and breakages ... Both are extremely good leaving groups due to resonance stabilization: S CH 3 O O SCH 3 O O O O 3. Solvent: The rate of the reaction can be affected ...
Strong Nucleophiles
tigerweb.towson.eduStrong Nucleophiles – • Usually anions with a full negative charge (easily recognizable by the presence of sodium, lithium or potassium counterions)
How Generational Theory Can Improve Teaching: Strategies ...
tigerweb.towson.eduCaulfields but as self-assured go-getters. Team-oriented Millennials have long worked in task groups and are skilled in collaborative effort. “From Barney and soccer to school uniforms and a new classroom empha-sis on group learning, Millennials are developing strong team instincts and tighter peer bonds,” write Strauss and Howe (2000, p. 44).
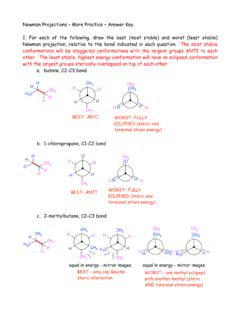
Newman Projections – More Practice- Answer Key
tigerweb.towson.eduNewman Projections – More Practice – Answer Key I. For each of the following, draw the best (most stable) and worst (least stable) Newman projection, relative to the bond indicated in each question. The most stable conformations will be staggered conformations with the largest groups ANTI to each ...
Related documents
Section 19.1. Acid-Base Buffer Solutions
christou.chem.ufl.edu** An acid-base buffer is a solution that lessens the change in [H 3O+] that would result when a strong acid or base is added ** A buffer is a concentrated solution of a weak acid (or base), together with a salt containing the conjugate base (or acid). How does a Buffer work? The Common-Ion Effect (example of Le Chatelier’s Principle)
Experiment 7 - Acid-Base Titrations
www.lahc.eduAcid + Base Salt + Water In this experiment, a phenolphthalein color indicator will be used. Phenolphthalein is colorless in acidic solutions and pink in basic solutions. Phenolphthalein is also used in forensic crime scene analysis to detect the presence of blood, Kastle-Meyer test. In the Kastle-Meyer test, hemoglobin catalyzes the
Acid-Base Chemistry
www.calstatela.eduThis is true of any conjugate pair of acid and base Salts of Acids and Bases ν When an acid and a base undergo an exchange reaction, the result is a salt and water: HX(aq) + MOH(aq) ↔ MX(aq) + H 2O acid base salt ν If a strong base is neutralized with a strong acid, the resulting solution contains only the salt HCl(aq) + NaOH(aq) ↔ NaCl ...
Base, Chemistry, Salt, Acid, Acid base chemistry, Acid base salt
Acid/Base Chemistry: Titration Lab
cpb-ca-c1.wpmucdn.comacid and a base react to produce a salt and water: In equation 1, the acid is HCl (hydrochloric acid) and the base is NaOH (sodium hydroxide). When the acid and base react, they form NaCl (sodium chloride), which is also known as table salt. The titration proceeds until the equivalence point is reached, where the number of moles of
Base, Chemistry, Salt, Acid, Titrations, Lab titration, Acid base chemistry
Acid-Base Titration Curves Using a pH Meter
web.gccaz.eduAcid Base Salt At the equivalence point for an acid-base neutralization reaction, the amount of base added is equal to the amount of acid initially present; thus, the acid has been completely neutralized. When a weak acid solution is initially present, the …
Using, Base, Salt, Acid, Titrations, Curves, Acid base titration curves using a, Acid base salt
Acid-Base Extraction
people.chem.umass.educarboxylic acid - high solubility in ether - low solubility in water salt of carboxylic acid - low solubility in ether - high solubility in water In the experiment done in this lab, a mixture of a carboxylic acid (stronger acid), a phenol (weaker acid), and a neutral compound will be separated by acid-base extractions. The separated compounds
Fluid, Electrolyte, and Acid-Base Balance
mymission.lamission.eduAcid-base balance Normal pH range of ECF is from 7.35 to 7.45. Most H+ comes from metabolism -- glycolysis, oxidation of fatty acids and amino acids, and hydrolysis of proteins. Homeostasis of pH in body fluids is regulated by acid-base buffer systems (primary control), respiratory centers in brain stem, and by kidney tubule secretion of H+
Acid-Base Physiology - PEDSCCM.org
pedsccm.orgAcid-Base Physiology Buffers H+ A-HCO3-CO2 Buffers H+ A-CO2 Cells Blood Kidney Lungs Fluids, Electrolytes, and Acid-Base Status in Critical Illness Laura Ibsen, M.D. Blood Gas Analysis--Insight into the Acid-Base status of the
The Common Ion Effect and Buffer Solutions
chemistry.armstrong.edu2 7 The Common Ion Effect and Buffer Solutions The general expression for the ionization of a weak monoproticacid is: The generalized ionization constant expression for a weak acid
Solutions, Acid, Common, Effect, Buffer, Common ion effect and buffer solutions