Tox considerations for oligonucleotide therapeutics
•Key considerations in preclinical testing - Pharmacological species restriction – Use of murine surrogate of the same ... Concentration-dependent prolongation of aPTT Single-dose, 4-hour Infusion 0.1 1 10 100 1000 0 1 4 4.25 4.5 5 8 12 16 28 52 76 100 168 340 Hours Post-dose 20K PEG-aptamer (ug oligo/mL) 25 35 45 55 65 75 aPTT (sec)
Tags:
Testing, Therapeutic, Considerations, Oligonucleotide, Tpat, Considerations for oligonucleotide therapeutics
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
I. Pulmonary Toxicology The Nanotoxicology Research ...
www.toxicology.org1 The Nanotoxicology Research Program in NIOSH Vincent Castranova, PhD NIOSH Nanotoxicology Research Program I. Pulmonary Toxicology (8 projects) II.
Research, Programs, Niosh, Nanotoxicology, The nanotoxicology research program in niosh
Nanotoxicology Specialty Section - Society of Toxicology
www.toxicology.org2011 Nanotoxicology Specialty SectionÑ293 members SOT Annual meetingÑ 2 workshops, 1 roundtable, 1 CE, 2 platforms, 4 poster sessions Pre-history 2011
Genotoxic impurities in small molecule drug candidates
www.toxicology.orgAssessment of Genotoxic Impurities in Small Molecule Drug Candidates John Wisler, PhD, DABT Kurt A Black, Ph.D, DABT Comparative Biology and Safety Sciences, Amgen Inc.
Drug, Impurities, Small, Molecules, Genotoxic, Genotoxic impurities in small molecule drug
SOT Abstract Submission Instructional Guide 2022
www.toxicology.org3 Enter the Abstract Submission Site Returning users: If you submitted a scientific session proposal or abstract for previous SOT Annual Meetings, you should use the same Login— usually your email address—and Password). Please note this system is separate from the SOT member section of the website and requires
Regulatory Toxicology - Society of Toxicology
www.toxicology.orgOverview/Objectives ... Duration of Repeat Dose Toxicology Studies Depends on Clinical Trial Duration . ICH M3 (R2) Non -Clinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorisation for Pharmaceuticals (2009) Industrial chemicals: Driven by production volume/import tonnage (ECHA 2014)
Regulatory, Clinical, Overview, Trail, Clinical trials, Authorisation
Quantitative Structure Activity Relationships: An overview
www.toxicology.org•Support prioritization in the absence of experimental data ... used as the test set. • Leave one out cross-validation: N models are developed each with (N − 1) chemicals as training set and 1 chemical ... Lack of proper chemical coverage in the training datasets which affects the applicability domain of the models and
An Update on FDA’s Good Laboratory Practice
www.toxicology.org–Published on August 24, 2016 (81 FR 58342) –Considered ANPRM comments and consistency with relevant OECD documents –Comment period closed on January 21, 2017 •90 day comment period •60 day extension –78 commenters –Multiple comments per submission www.fda.gov
Related documents
Anti-phospholipid Antibody Testing (Lupus Anticoagulant ...
clevelandcliniclabs.comfor APA testing. Because the antigen target of ACAs is B2GP1 bound to cardiolipin, B2GP1 antibodies are considered to be more specific than ACA assays. Clinical Indications for Testing Suspicion for APS in patients with an elevated aPTT, unexplained thrombocytopenia, or a history of arterial and venous thrombosis and/or obstetric complications.
The Coagulation Cascade - Deranged Physiology
derangedphysiology.comTesting the PT is a way of looking at the function of the Vitamin K-dependent enzymes; namely II, VII and X . Factor IX is also vitamin-K dependent, but is not tested. INR: international normalized ratio . Comparison of a given PT to an average PT. An INR of 2 means blood is clotting twice as slowly as normal. aPTT: Activated Partial ...
Guideline Retningslinjer for antitrombotisk behandling og ...
files.magicapp.orgApr 08, 2015 · APTT = 70-90 sekunder forenlig medhøy plasmakonsentrasjon APTT > 90 sekunder forenlig med overdosering INR > 1,5 forenlig med overdosering (sykehusmetoder) Tolkning av APTT og INR ved behandling med apixaban og rivaroksaban Råd (Ikke Gradert) Eller Informasjon Usikker nytte av APTT og INR.
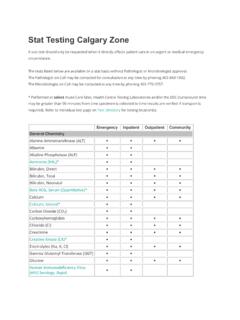
Stat Testing Calgary Zone - Alberta Health Services
www.albertahealthservices.caStat Testing Calgary Zone A stat test should only be requested when it directly affects patient care in an urgent or medical emergency circumstance. The tests listed below are available on a stat basis without Pathologist or Microbiologist approval. The Pathologist on Call may be contacted for consultation at any time by phoning 403-860-1802.
Test Selection Guide - Dorevitch
www.dorevitch.com.au• Alpha thalassaemia genetic testing • Beta thalassaemia genetic testing • Factor V Leiden • Prothrombin G20210A • Haemochromatosis (C282Y, H63D & S65C) Haematological Oncology • BCR/ABL • FLT3 & NPM1 • Janus Kinase (JAK2) • Calreticulin (CALR) • B-cell IgH gene rearrangement • T-cell Receptor gene rearrangement • Bcl-1 ...
COVID-19 patients: Anticoagulation Management at UNC ...
www.med.unc.edu• Test both, aPTT and “Heparin-unfractionated” (anti-Xa) test for first 48 hrs • assess how well these two tests correlate • If good correlation, then use aPTT; if poor correlation: use “Heparin-unfractionated” (anti-Xa) test. UFH monitoring: UFH 80 U/kg bolus, then 18 U/kg/hr •Target “hep correlation” or anti-Xa: 0.3-0.7 U/ml
GUIDE TO PATHOLOGY TESTS - Clinical Labs
www.clinicallabs.com.auANTENATAL TESTING Note: "Antenatal Screen" is no longer a valid test request. Please specify which of the tests below are requested. FBE, ECU, LFT, TFT, Blood group and antibodies, First visit and < 20 weeks: Fasting glucose, Rubella Ab status, Hep B surface Antigen and Hep C antibody, HIV serology, Syphilis serology, Urine MCS, Cervical Cytology