Search results with tag "Alkynes"
alkenes and alkynes worksheet and key02 25 10
www.saddleback.eduAlkenes and Alkynes Worksheet and Key 1. Draw the line bond structures for the following alkenes, cyclic alkenes, and alkynes: a) alkenes that contain 4 carbon atoms (three possible) b) cyclic alkenes that contain 4 carbon atoms (three possible)
Synthesis of Acetylene - ChemConnections
chemconnections.orgAlkynes → trans-Alkenes Metal-Ammonia Reduction of Alkynes RCH 2CH2R' Another way to convert alkynes to alkenes is by reduction with sodium (or lithium or potassium) in ammonia. trans-Alkenes are formed. RC CR' RCH CHR' Partial Reduction CH3CH2 H CH2CH3 H (82%) CH3CH2C CCH2CH3 C C Na, NH3 Example
Introduction to Alkenes and Alkynes In an alkane, all ...
utdallas.eduIntroduction to Alkenes and Alkynes! In an alkane, all covalent bonds between carbon were σ" (σ bonds are defined as bonds where the electron density!
NOMENCLATURE IN ORGANIC CHEMISTRY
scilearn.sydney.edu.au(ii) Alkenes 5 A. One double bond 5 B. More than one double bond 5 C. E/Z Isomers in Alkenes 6 (iii) Alkynes 8 (iv) Combined Alkenes and Alkynes 8 (v) Cyclic Hydrocarbons 9 3. COMPOUNDS CONTAINING HALOGENS AND NITRO GROUPS 10 4. COMPOUNDS WITH FUNCTIONAL GROUPS NAMED AS SUFFIXES 12 (i) General Naming Scheme 12
8. CHEMISTRY (Code No. 043) Rationale
cbseacademic.nic.inAlkynes - Nomenclature, structure of triple bond (ethyne), physical properties, methods of preparation, chemical reactions: acidic character of alkynes, addition reaction of - hydrogen, halogens, hydrogen halides and water.
II Alkanes, Alkenes, and Alkynes
web.chemistry.gatech.edu1 II Alkanes, Alkenes, and Alkynes Hydrocarbon : Compound composed of only carbon and hydrogen Saturated Hydrocarbons : Compound with only single bonds
PRACTICE EXERCISE – ORGANIC CHEMISTRY I Alkynes …
personal.utdallas.eduAlkynes Synthesis and Reactions FOR QUESTIONS 1-4, DRAW A LEWIS OR LINE-ANGLE FORMULA AND GIVE THE IUPAC NAME. 1) (CH3)2C(CH2CH3)CCCH(CH3)2 2) HCCCH2CH2CH3 3) CH3CH=CHCH=CHCCCH3 4) BrCH2CH2CCCH2CH3 5) Draw acetylene 6) Draw (S)-5-phenyloct-2-yne 7) Draw hepta-3,6-dien-1-yne 8) The carbon-carbon triple bond of …
Naming Alkynes Worksheet #1 (C4 305)
msgalloway.infoNaming Alkynes Worksheet #2 Draw the structural formula for each of the following: 3-ethyl-l-pentyne 3,3-dimethyl- l -butyne 4,4,5 — timethyl-2-hexyne
Carboxylic Acids - Rutgers University
crab.rutgers.eduCleavage of Alkynes and Alkenes Alkenes react with concentrated KmnO 4 to produce intermediate glycols which react further to produce either carboxylic acids or ketones (depending on the original alkene substituents). E.g. Alkynes also react with conc. KMnO 4 to give carboxylic acids, and the same transformation can be achieved by the use of ...
from Organic Chemistry
chemistry.ucr.edu2. Alkanes and Cycloalkanes 3. Haloalkanes, Alcohols, Ethers, and Amines 4. Stereochemistry 5. Organic Spectrometry II. Reactions, Mechanisms, Multiple Bonds 6. Organic Reactions *(Not yet Posted) 7. Reactions of Haloalkanes, Alcohols, and Amines. Nucleophilic Substitution 8. Alkenes and Alkynes 9. Formation of Alkenes and Alkynes. Elimination ...
Laboratory 21: Properties of Alkanes, Alkenes, and Alkynes ...
www.chemhaven.orgLaboratory 21: Properties of Alkanes, Alkenes, and Alkynes B. Physical Properties When a compound burns in the presence of oxygen the reaction is called a combustion.
INTRODUCTION TO ORGANIC NOMENCLATURE
utdallas.eduOVERVIEW OF FUNCTIONAL GROUPS BASED ON ATOM HYBRIDIZATION I. HYDROCARBONS - Substances containing only carbon and hydrogen. ALKANES C C Only sp3 carbon present (technically not a functional group) ALKENES C C At least one π-bond between two sp2 carbons present ALKYNES C C At least one triple bond between two sp carbons present II…
NOMENCLATURE OF ORGANIC COMPOUNDS
www.chymist.comAlkynes contain at least one carbon to carbon triple bond. The suffix used is –yne. 6 Naming is the same as used for alkanes, except that the parent structure is the longest continuous chain of carbon atoms that contains the carbon-carbon double bond or triple bond. The name is
Get In The Zone The basics of Reading Infrared ...
www.chem.ucla.eduN i t r i l e ( C ≡ N ) One around 2260‐2220 cm‐1 Alkynes have variable and sharp peaks, such as the peak in
The GC Column - Agilent
www.agilent.combut also contain unsaturated bonds (Alkenes, alkynes and aromatic compounds) Selectivity Interactions Group/Presentation Title Agilent Restricted Page 24 •Dispersion •Dipole •Hydrogen bonding. Dispersion Interaction DH vap Group/Presentation Title Agilent Restricted Page 25 •Separation by differences in analyte heat of
Experiment #3 - Hydrocarbons
employees.oneonta.eduAlkenes, alkynes, aromatic compounds, and cyclic alkanes are unsaturated because hydrogen can be added to them, in theory and usually in practice, making them into acyclic alkanes. Some examples follow. Physical Properties Some molecules carry an electrical charge because there is a difference between
Bromine Water Test - organicchem.org
organicchem.orgBromine Water Test Functional Group(s): Alkenes, alkynes, Phenols, enols Known(s): 1-butanol, 2-butanol, tert-butyl alcohol, phenol, decene Procedure Set up 6 small (12 X 75mm) test tubes in a test tube rack in the hood. Label the test tubes #1-6.
B.Sc. - FIRST YEAR - mjpru.ac.in
www.mjpru.ac.informulae, Fischer and flying wedge formulae, Difference between configuration and conformation. Unit – III V. Alkenes, Cycloalkenes, Dienes and Alkynes:
Chemistry - Chhatrapati Shahu Ji Maharaj University, Kanpur
www.kanpuruniversity.orgformulae, Fischer and flying wedge formulae, Difference between configuration and conformation. Unit – III V. Alkenes, Cycloalkenes, Dienes and Alkynes:
B.Sc. - FIRST YEAR - M. J. P. Rohilkhand University
mjpru.ac.informulae, Fischer and flying wedge formulae, Difference between configuration and conformation. Unit – III V. Alkenes, Cycloalkenes, Dienes and Alkynes:
GC Column Selection Guide - Sigma-Aldrich
www.sigmaaldrich.comPolarizable compounds are compounds composed of carbon and hydrogen, but contain one or more double or triple carbon-carbon bonds. These compounds include alkenes, alkynes, and aromatic (benzene-ring containing) hydrocarbons. Highly polar capillary columns are generally used to separate these compounds. Phase Polarity Based on Compound Polarity
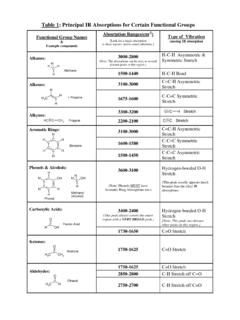
Table 1: Principal IR Absorptions for Certain Functional ...
academics.wellesley.eduAlkynes: HCC CH 3 Propyne 2200-2100 CCStretch 3100-3000 C=C-H Asymmetric Stretch 1600-1580 C-C=C Symmetric Stretch Aromatic Rings: C C C C C C H H H H H H Benzene 1500-1450 C-C=C Asymmetric Stretch Phenols & Alcohols: C C C C C C H H H H H OH C H OH H H Phenol Methanol (Alcohol) 3600-3100 (Note: Phenols MUST have Aromatic Ring Absorptions too ...
1H NMR Tables - udel.edu
www1.udel.eduSubstituted Alkynes and CEN 2.10 0.97 1.50 c Experimental spa Il-I chemical shifts (ppm) (cont.). Functionalized Alkanes 70 2.09 05 1.57 1.69 56 0.97 1.65 PhO 3.86 1.53 HO 3.49 0.11 1.56 AcO 3.96 2.42 0.91 0.97 1.76 2.88 0.93 354 0.89 1.68 2.14 151 HO 0.93 2.32 1.43 2.31 2.01 3.67 MeO 1.00 1.00 2.29 1.06 0.95 1.89 1.06 4.28 1.77 1.38 1.81 1.88 3.18
Chapter 8 - Alkenes, Alkynes and Aromatic Compounds
wou.edu2 H 4, whereas that for ethyne is C 2 H 2. Thus, until you become more familiar the language of organic chemistry, it is often most useful to draw out line or partially-condensed structures, as shown below: (Back to the Top) 8.2 Properties of Alkenes The physical properties of alkenes are similar to those of thealkanes. Table 8.1 shows
pKa Chart 1 2 conjugate acid conjugate base conjugate acid ...
cactus.dixie.edualkynes 26 ketones esters (enolates) 18-21 25 hydrogen cyanide 9.1 tertiary alcohols phenols 10 1,3-diketones 9 10.5 10.6 15.7 protonated amines water cyclopentadiene amides (aromatic) 15 15 conjugate acid conjugate base conjugate acid conjugate base (hydroxide) (tert-butoxide) (ester enolates) s t r o n g e s t b a s e s w e a k e s t a c i d ...
IR Spectroscopy by Functional Group
users.wfu.eduIR Spectroscopy by Functional Group Alkanes Indentification of absorbance Wavenumbers, cm-1 sp3 C-H stretch 2960-2850 (<3000) Alkenes sp2 C-H stretch 3100-3020 cm-1 (>3000) C=C stretch 1680-1620 cm-1 (often weak) Alkynes sp C-H stretch 3333-3267 cm-1 C-C triple bond stretch 2260-2100 cm-1 (often weak) Nitriles
Naming Rules for Organic Compounds
web.viu.caAlkynes - compounds containing carbon-carbon triple bonds 1) Are named and numbered like alkenes. Add "yne" to root name. Aromatic Hydrocarbons - Compounds containing benzene-like rings 1) Benzene is the simplest aromatic compound containing cyclic structure of six carbons with a total of three double bonds. benzene NOT 1,3,5-cyclohexatriene ...
HYDROCARBONS 365 - NCERT
www.ncert.nic.inplasticine balls for atoms. For alkenes, alkynes and aromatic hydrocarbons, spring models can be constructed. 13.2 ALKANES As already mentioned, alkanes are saturated open chain hydrocarbons containing carbon - carbon single bonds. Methane (CH 4) is the first member of this family. Methane is a gas found in coal mines and marshy places. If
CHAPTER 7 ALCOHOLS, THIOLS, PHENOLS, ETHERS
www.siue.eduhigher priority. Thus many compounds have the alcohol parent system. The IUPAC system names the longest chain hydrocarbon as the parent, drops the ending -e and adds a new ending of -ol. The alcohol has a higher priority in nomenclature than alkenes, alkynes, alkyl groups and halogen and is given a number lower than any of those groups.
NOMENCLATURE OF ORGANIC COMPOUNDS
chymist.comAlkynes contain at least one carbon to carbon triple bond. The suffix used is –yne. 6 Naming is the same as used for alkanes, except that the parent structure is the longest continuous chain of carbon atoms that contains the carbon-carbon double bond or triple bond. The name is
Functional GroupsFunctional Groups
www.csus.eduAlkynes have a C-C triple bondtriple bond ... aromatic ringaromatic ring. note Aldehyde C-H stretches at 2800-2700 cm-1. IR of Ketones and Esters
Functional Groups - Purdue University
www.chem.purdue.eduC. Alkynes • contain a carbon-carbon triple bond . 2 D. Arenes • contain a benzene group E. Alcohols • contain an –OH (“hydroxyl”) group bonded to a tetrahedral carbon atom F. Ethers • contain an oxygen atom bonded to two carbon atom groups by single bonds . 3
Handout: Naming Organic Compounds
laney.eduAlkynes Parent is longest carbon chain containing the double or triple bond. Parent name starts with position number of multiple bond. May need cis/trans designation. –ene ... Aromatic Nomenclature (*Functional group priority is same in aromatic and aliphatic nomenclature.)
5-Reactions of Hydrocarbons - Laney College
laney.eduJan 05, 2012 · Alkenes and alkynes are much more reactive than alkanes. They will react readily with Br 2 or Cl 2, and ultraviolet light is not needed for the reaction. This reaction is an addition reaction – the halogen atoms will add at the site of the double bond only. Therefore, this reaction is selective– only one product will result.
Worksheets for Organic Chemistry - cffet.net
www.cffet.netChemistry of Natural Substances – Organic Chemistry Worksheets 7 Worksheet 4 Alkenes/Alkynes Question 1. Predict the products of the reaction of 2-methyl-2-pentene with each of the following.Name each product. a) HBr b) dilute sulfuric acid c) Br
Similar queries
Alkenes and Alkynes Worksheet and, Alkynes, Introduction to Alkenes and Alkynes, Bonds, Alkenes, Compounds, Properties, And Alkynes, Alkanes, Alkenes and alkynes, Laboratory 21: Properties of Alkanes, Alkenes, and Alkynes, NOMENCLATURE, Infrared, Alkynes and aromatic, Aromatic compounds, Bromine, Chemistry, Sigma-Aldrich, And aromatic, Alkenes, Alkynes and Aromatic Compounds, Of alkenes, Aromatic, HYDROCARBONS 365, Handout: Naming Organic Compounds, 5-Reactions of Hydrocarbons, Worksheets for Organic Chemistry