Improving the reporting of pragmatic trials: an …
research methods & reporting Randomised controlled trials are used to assess the ben ‑ efits and harms of interventions in health care. If con‑
Tags:
Reporting, Improving, Trail, Pragmatic, Improving the reporting of pragmatic trials
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
research methods & reporting - consort …
www.consort-statement.orgPage 1 of 28 BMJ | online FiRST | bmj.com research methods & reporting “The whole of medicine depends on the transparent report ‑ ing of clinical trials.”1 Well designed and properly executed randomised con‑
CONSORT 2010 Statement CONSORT 2010 …
www.consort-statement.orgCONSORT 2010 Statement 2 www.thelancet.com Published online March 24, 2010 Webappendix Indeed, CONSORT Group members continually monitor the literature. Information gleaned from these
Testament, 2010, Consort 2010 statement consort 2010, Consort, Consort 2010 statement
Meta-analysis of Observational Studies in …
www.consort-statement.orgCONSENSUS STATEMENT Meta-analysis of Observational Studies in Epidemiology A Proposal for Reporting Donna F. Stroup, PhD, MSc Jesse A. Berlin, ScD
Analysis, Team, Studies, Observational, Meta analysis of observational studies
research methods & reporting - CONSORT Statement
www.consort-statement.orgresearch methods & reporting BMJ statement. CONSORT statement. randomised controlled • • • •
Research, Methods, Reporting, Research methods amp reporting
Using the CONSORT for Abstracts checklist: some …
www.consort-statement.orgUsing the CONSORT for Abstracts checklist: some examples This is a series of slides providing illustrative examples of raiding illustrative examples of randomized ndomized
Checklist, Example, Some, Abstracts, Consort, Consort for abstracts checklist, Some examples
Item Reported No Checklist item on page No - …
www.consort-statement.orgCONSORT 2010 checklist Page 1 CONSORT 2010 checklist of information to include when reporting a randomised trial* Section/Topic Item No Checklist item
Time, Checklist, Reported, Consort, Item reported no checklist item on
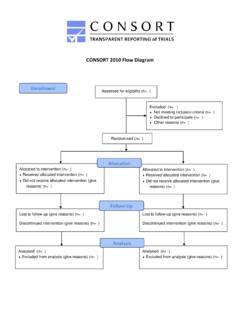
CONSORT 2010 Flow Diagram
www.consort-statement.orgCONSORT 2010 Flow Diagram Assessed for eligibility (n= ) Excluded (n= ) Not meeting inclusion criteria (n= ) Declined to participate (n= ) Other reasons (n= )
CONSORT 2010 Statement: updated guidelines for …
www.consort-statement.orgOpen Medicine 2010;4(1):e61 Review Schultz et al.
Declaración CONSORT 2010 ... - consort-statement.org
www.consort-statement.orgTabla 1 CONSORT 2010. Lista de comprobacio´n de la informacio´n que hay que incluir al comunicar un ensayo clı´nico aleatorizado* Seccio´n/tema I´tem n8 I´tem de la lista de comprobacio´n Informado en
Liste de contrôle CONSORT 2010 à inclure pour rendre ...
www.consort-statement.orgListe de contrôle CONSORT 2010 Page 1/2 Liste de contrôle CONSORT 2010 à inclure pour rendre compte d’un essai randomisé* Section/sujet
Related documents
PHASES OF CLINICAL TRIALS - University of Virginia
www.virginia.eduPHASES OF CLINICAL TRIALS Four phases of clinical trials and medicine development exist and are defined below. Each of these definitions is a functional one and the terms are not defined on a strict
Virginia, University, Clinical, Trail, Clinical trials, University of virginia
PHS Human Subjects and Clinical Trials Information
medschool.ucla.eduPHS Human Subjects and Clinical Trials Information OMB Number: 0925-0001 Expiration Date: 03/31/2020 Please complete the human subjects section of the Research & Related Other Project Information form prior to completing this form.
Information, Clinical, Subject, Trail, Subjects and clinical trials information
Vol. 9, No. 2, February 2013 Happy Trials to You
www.chubb.comVol. 9, No. 2, February 2013 “Happy Trials to You” Subscribe free at www.firstclinical.com © 2013 First Clinical Research and the Author(s)
Bacterial vaginosis: a review on clinical trials with ...
www.newmicrobiologica.orgNEw MICRoBIoLogICA, 36, 229-238, 2013 Bacterial vaginosis: a review on clinical trials with probiotics Paola Mastromarino 1, Beatrice Vitali2, Luciana Mosca 3 1Department of Public Health and Infectious Diseases, Section of Microbiology, Roma, Italy; 2Department of Pharmacy and Biotechnology, University of Bologna, Bologna, Italy; 3Department of …
“No body” Murder Trials in the United States© …
www.nobodycases.com{888 / 95 / 00077099.DOC v1} Defendant’s Victim’s Name Trial Date Location Result Adamson, Stacey Daniel Jamison April 2004 Schenectady, NY Defendant and two others
Clinical trials appendix Q1 2018 Results update
www.astrazeneca.com4 Table of contents slide Movement since Q42017 results announcement Q1 2018 New Molecular Entity (NME) pipeline Q1 2018 Lifecycle Management (LCM) pipeline
COMMONLY USED ABBREVIATIONS AND TERMS …
tracs.unc.eduADR . Drug Reaction : Adverse AE . Adverse Event : AUC . Area Under the Curve : BLA . Biologic Licensing Application : BUN . Blood Urea Nitrogen : CAP . College of ...
Terms, Used, Commonly, Abbreviations, Commonly used abbreviations and terms